Taletrectinib in TKI-Pretreated Patients With ROS1+ Non-Small Cell Lung Cancer: Updated Data From TRUST-I and TRUST-II
Geoffrey Liu,1 Jorge Nieva,2 Lyudmila Bazhenova,3 Chang-Min Choi,4 Qitao Yu,5 Shunichi Sugawara,6 Noriko Yanagitani,7 Filippo de Braud,8 Huijie Fan,9 Enriqueta Felip,10 Emilio Bria,11 Maurice Pérol,12 Feiwu Ran,13 Wei Wang,13 Xianyu Zhang,13 Wenfeng Chen,13 Caicun Zhou14
1Princess Margaret Cancer Centre, Temerty School of Medicine, University of Toronto, Toronto, ON, Canada; 2Norris Comprehensive Cancer Center, University of Southern California, Los Angeles, CA, USA; 3UC San Diego Moores Cancer Center, San Diego, CA, USA;
4Asan Medical Center, University of Ulsan College of Medicine, Seoul, Republic of Korea; 5Affiliated Tumor Hospital of Guangxi Medical University, Nanning, China; 6Sendai Kousei Hospital, Sendai, Japan; 7Cancer Institute Hospital of Japanese Foundation for Cancer Research, Tokyo, Japan;
8University of Milan, Milan, Italy; 9The First Affiliated Hospital of Zhengzhou University, Zhengzhou, China; 10Vall d’Hebron University Hospital, Barcelona, Spain; 11Fondazione Policlinico Universitario Agostino Gemelli IRCCS, Università Cattolica del Sacro Cuore, Rome, Italy;
12Léon Bérard Cancer Center, Lyon, France; 13Nuvation Bio Inc., New York, NY, USA; 14Shanghai East Hospital and Thoracic Cancer Institute, Tongji University School of Medicine, Shanghai, China
Presented at the American Association for Cancer Research (AACR) Annual Meeting | April 17–22, 2026 | San Diego, CA, USA
For more information, please contact Dr Geoffrey Liu: geoffrey.liu@uhn.ca
Jump to a section:
Background
- Taletrectinib is a next-generation, CNS-active, selective ROS1 TKI approved in the US, Japan, and China for the treatment of patients with locally advanced or metastatic ROS1+ NSCLC1–5
- In the Phase 2 TRUST-I (NCT04395677) and TRUST-II (NCT04919811) studies, taletrectinib demonstrated robust efficacy, including IC activity and efficacy against G2032R mutations, with a manageable safety profile in TKI-naïve and TKI-pretreated patients with ROS1+ NSCLC6–8
- Here we report updated efficacy data in TKI-pretreated patients from TRUST-I and TRUST-II, and updated data from an integrated safety analysis
Methods
- The study designs of TRUST-I and TRUST-II have been previously reported6
- The efficacy populations reported here include TKI-pretreated patients from TRUST-I and TRUST-II with ≥1 measurable lesion at baseline per RECIST v1.1 by IRC who started treatment on taletrectinib 600 mg QD
- The safety population includes TKI-naïve and TKI-pretreated patients with ROS1+ NSCLC from Phase 1 and Phase 2 studies who received ≥1 dose of taletrectinib 600 mg QD
Efficacy in TKI-Pretreated Patients
Demographics and Baseline Characteristics for TKI-Pretreated Patientsa
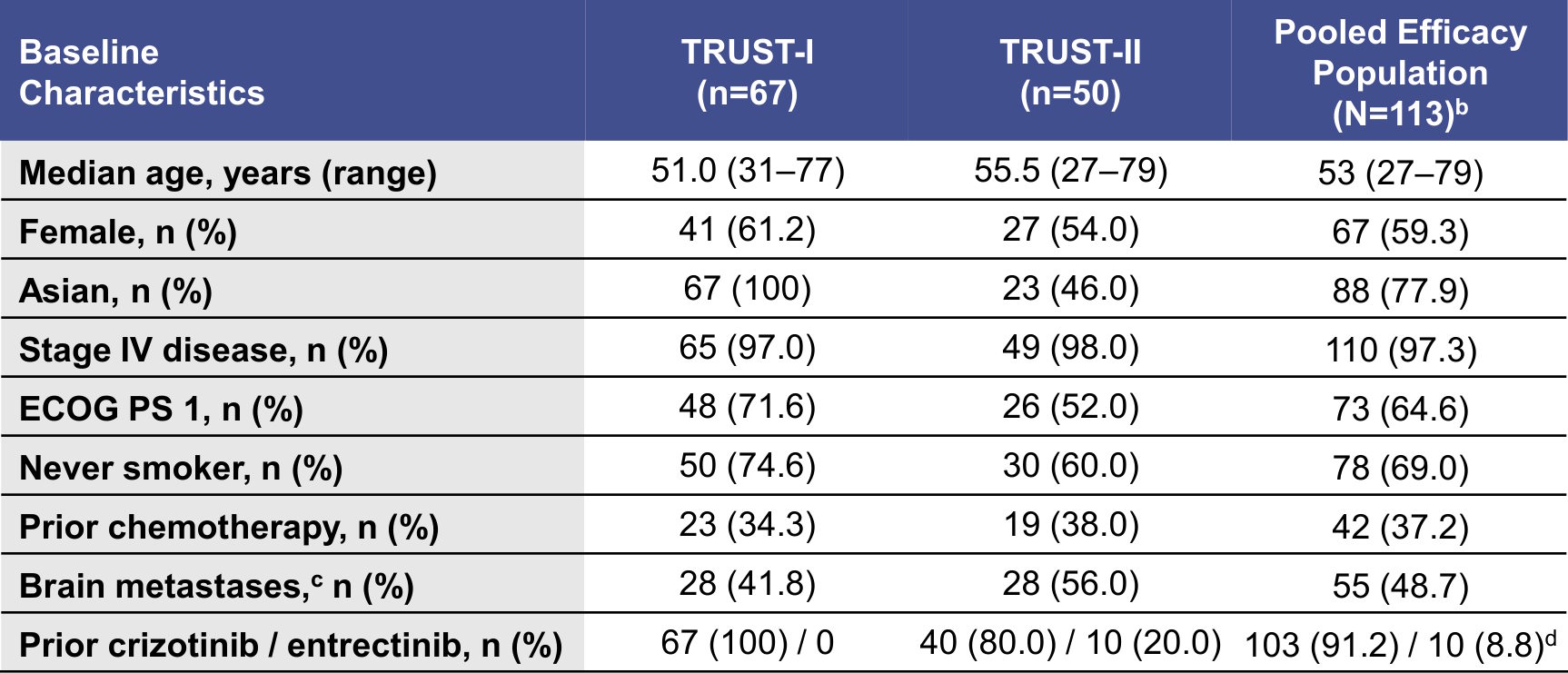
aPatients from TRUST-I and the registrational cohort 2 of TRUST-II who received one prior TKI. bIncludes patients with ≥1 measurable baseline lesion per RECIST v1.1 by IRC. cAssessed by IRC per mRECIST v1.1. dTwo patients were enrolled following crizotinib intolerance and 111 patients were enrolled following PD on a prior TKI.
Tumor Response in TKI-Pretreated Patients
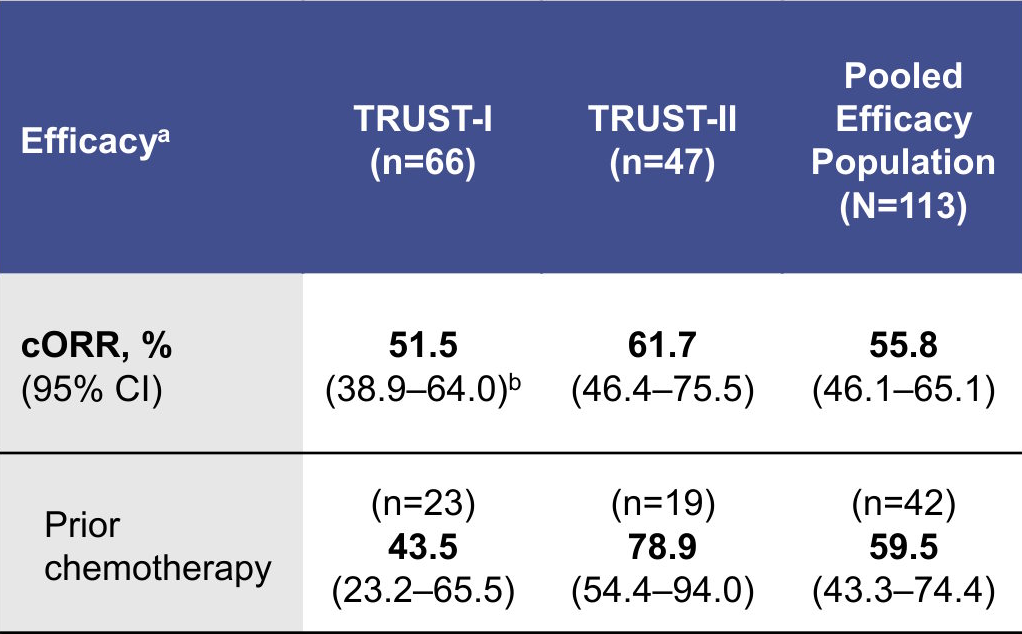
IC efficacy data are shown in the Supplement
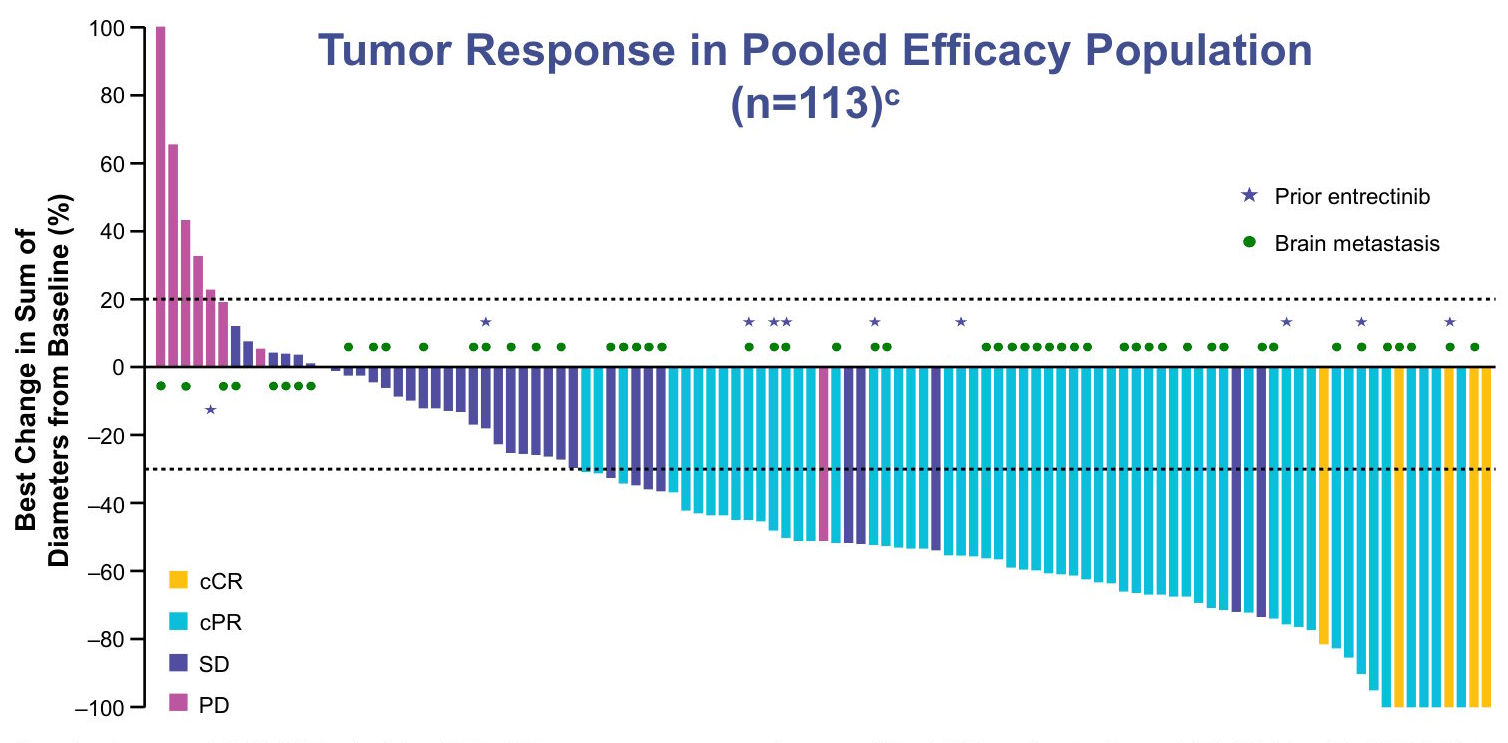
Data cutoff: August 31, 2025. aAssessed in patients with ≥1 measurable baseline lesion per RECIST v1.1 by IRC. bResponses were observed in 8/12 patients from TRUST-I with G2032Rmutations (ORR 66.7% [95% CI: 34.9–90.1]). cOne patient had a change of 136.5%, which is truncated at 100%. Six patients with cBOR of NE are not shown in the figure.
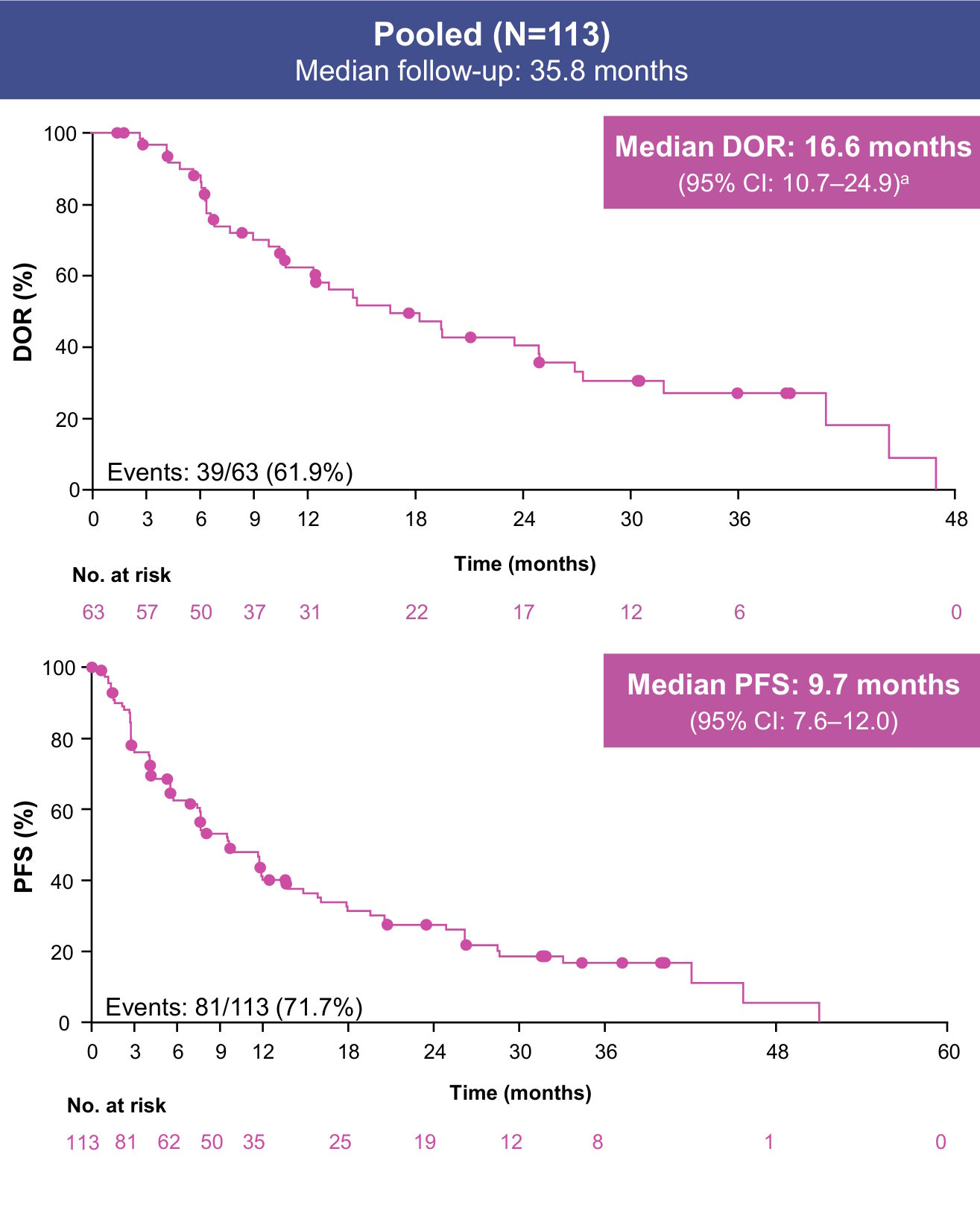
Data cutoff: August 31, 2025. aDOR reported only for patients with a CR or PR.
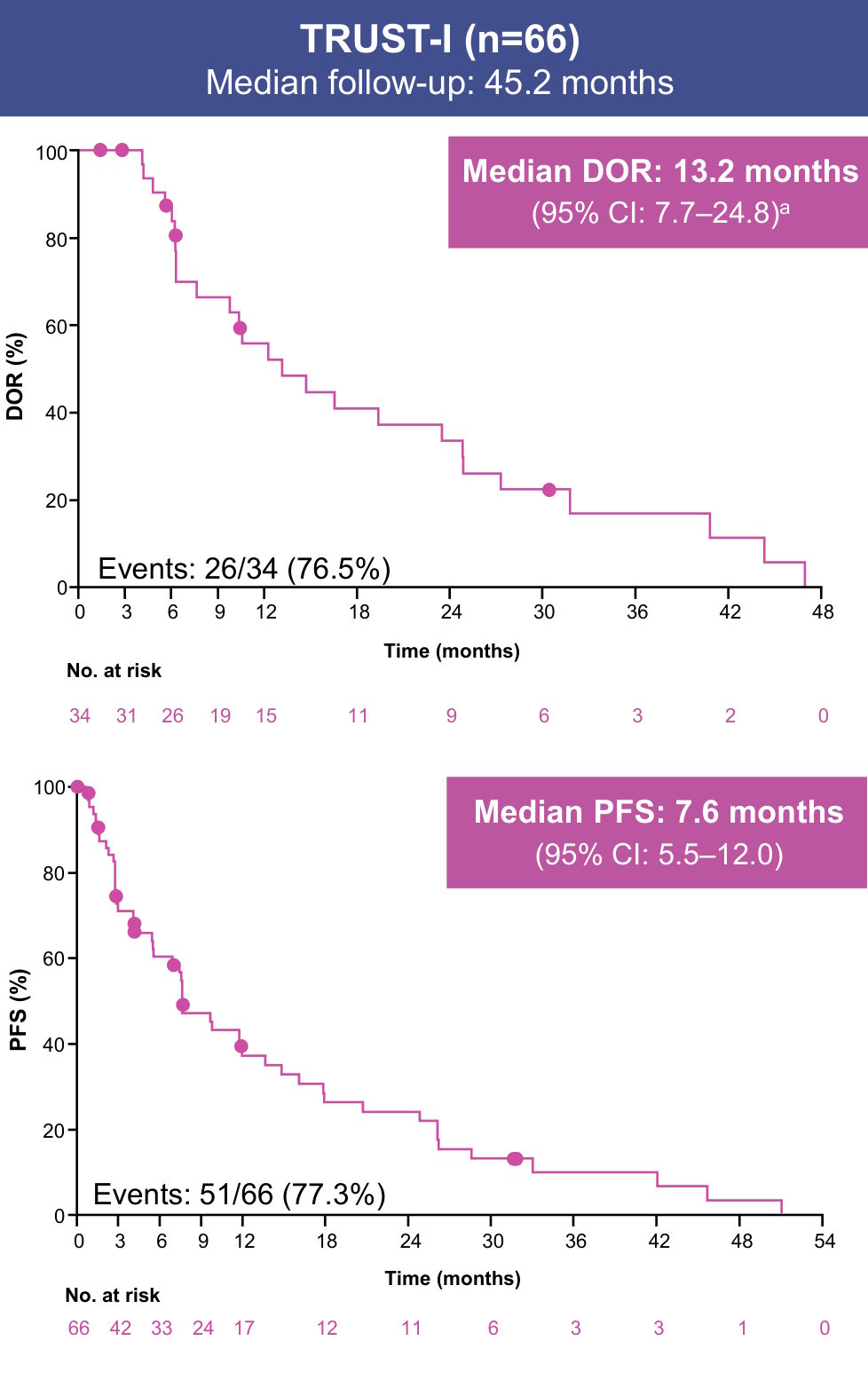
Data cutoff: August 31, 2025. aDOR reported only for patients with a CR or PR.
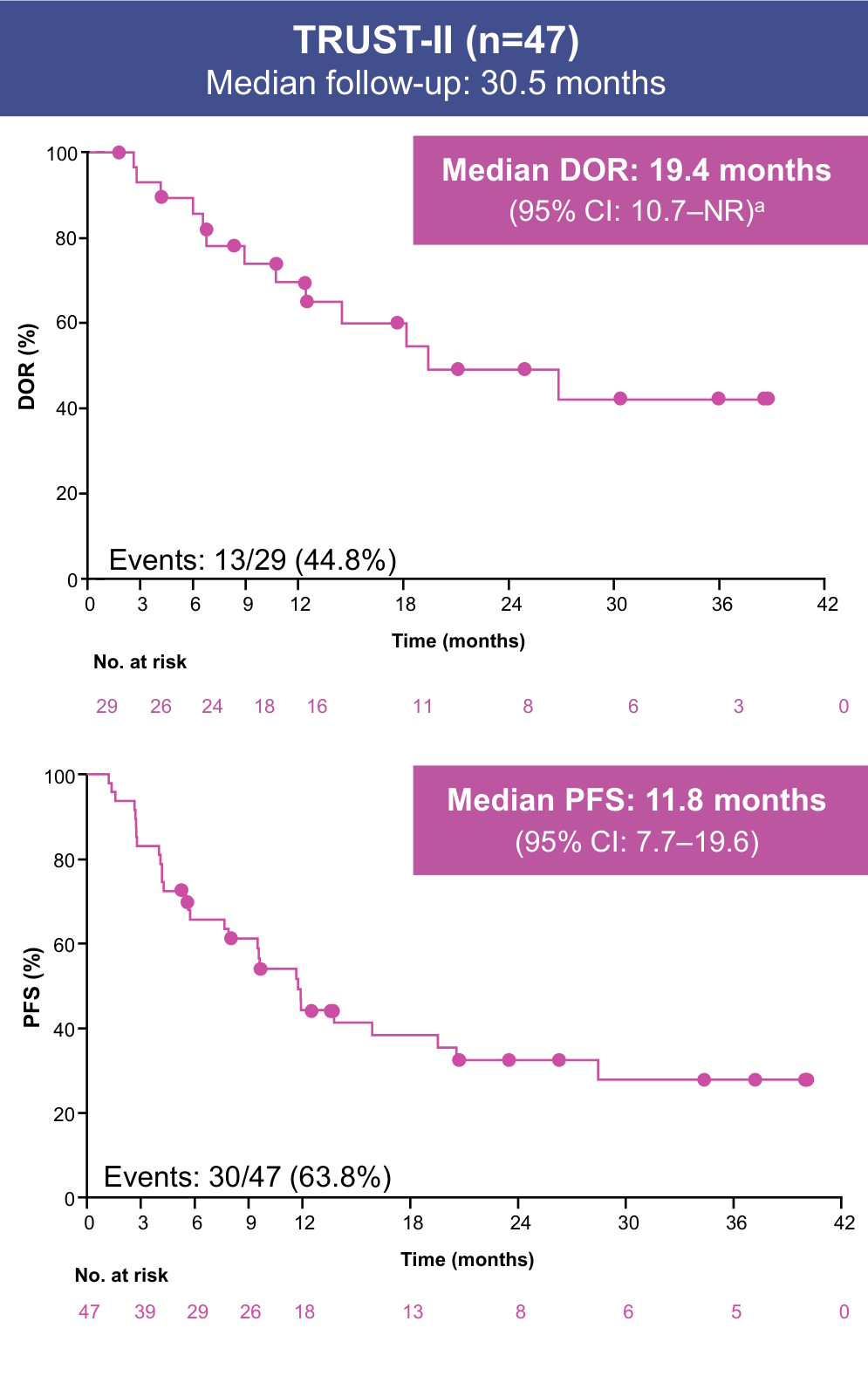
Data cutoff: August 31, 2025. aDOR reported only for patients with a CR or PR.
Median OS (95% CI) was 29.8 months (23.2–46.0) in the pooled efficacy population, 25.6 months (19.2–31.9) in TRUST-I, and NR (23.2–NR) in TRUST-II (Supplement)
Integrated Safety Analysis (N=363)
- As of August 31, 2025, the integrated safety population included 363 TKI-naïve and TKI-pretreated patients with ROS1+ NSCLC from Phase 1 and Phase 2 studies who received ≥1 dose of taletrectinib 600 mg QD (Supplement)
- With longer follow-up, no new safety signals were identified, and safety was consistent between the integrated safety population and TKI-pretreated patients (Supplement)
- The most common (any-grade) TEAEs were increased AST, increased ALT, diarrhea, nausea, and vomiting (Supplement)
- TEAEs led to dose interruptions in 42.7% of patients, dose reductions in 31.3%, and treatment discontinuations in 8.5%
Conclusions
- With ~3 years of follow-up in the pooled analysis of TRUST-I and TRUST-II, taletrectinib maintained durable responses in TKI-pretreated patients with ROS1+ NSCLC
- With longer follow-up, taletrectinib demonstrated a manageable safety profile, with no new safety signals
- These data support taletrectinib as an effective and tolerable treatment option for patients with ROS1+ NSCLC after prior TKI therapy
Abbreviations
ALT, alanine aminotransferase; AST, aspartate aminotransferase; BOR, best overall response; c, confirmed; CI, confidence interval; CNS, central nervous system; CR, complete response; DOR, duration of response; ECOG PS, Eastern Cooperative Oncology Group performance status; IC, intracranial; IRC, independent review committee; (m)RECIST v1.1, (modified) Response Evaluation Criteria in Solid Tumors version 1.1; NE, not evaluable; NR, not reached; NSCLC, non-small cell lung cancer; ORR, objective response rate; OS, overall survival; PD, progressive disease; PFS, progression-free survival; PR, partial response; QD, once daily; ROS1, ROS proto-oncogene 1; SD, stable disease; TEAE, treatment-emergent adverse event; TKI, tyrosine kinase inhibitor; US, United States
References
- Katayama R, et al. Nat Commun 2019;10:3604
- Nagasaka M, et al. Future Oncol 2023;19:123–135
- IBTROZI® (taletrectinib). Prescribing Information. Nuvation Bio Inc. 2025
- Nippon Kayaku. IBTROZI® Capsules 200mg (taletrectinib) has been approved in Japan. Accessed March 27, 2026; https://www.nipponkayaku.co.jp/english/news/detail.php?n=20250919_6G5AI1Y7
- Nuvation Bio. Nuvation Bio Receives Approval from China’s NMPA for Taletrectinib. Accessed March 27, 2026; https://investors.nuvationbio.com/news/news-details/2025/Nuvation-Bio-Receives-Approval-from-Chinas-National-Medical-Products-Administration-for-Taletrectinib-for-Patients-with-Advanced-ROS1-positive-Non-Small-Cell-Lung-Cancer
- Pérol M, et al. J Clin Oncol 2025;43:1920–1929
- Li W, et al. J Thorac Oncol 2025;20(Suppl 1):S500
- Liu G, et al. J Thorac Oncol 2025;20(Suppl 1):S56
Acknowledgments
We would like to thank all patients who participated in these studies, the study investigators, and their staff. These studies were sponsored by Nuvation Bio Inc. Medical writing support was provided by Flaminia Fenoaltea, MSc, of Ashfield MedComms, an Inizio company, and was funded by Nuvation Bio Inc.