Taletrectinib, a next-generation selective ROS1 inhibitor, inhibits TRKB signaling-induced invasive potential of lung cancer cells
Nuvation Bio, Inc., New York, NY
Presented at the American Association for Cancer Research (AACR) Annual Meeting | April 17–22, 2026 | San Diego, CA, USAJump to a section:
Background
- ROS1 fusions are present in approximately 2% of patients with non–small cell lung cancer (NSCLC), driving cancer cell growth and survival.1
- FDA-approved tyrosine kinase inhibitors (TKIs) for ROS1 fusion-positive NSCLC include crizotinib, entrectinib, repotrectinib and more recently, taletrectinib.1
- Taletrectinib is a potent, CNS-active, selective, next-generation ROS1 TKI for the treatment of patients with locally advanced or metastatic ROS1-positive NSCLC.
- Taletrectinib is currently approved by the FDA and is also approved in China and Japan; additionally, the EMA validated the Marketing Authorization Application for taletrectinib in Europe.
- Among patients with advanced ROS1-positive NSCLC as of August 31, 2025, pooled results from the TRUST-I and TRUST-II studies of taletrectinib demonstrated2:
- TKI-naïve: a confirmed objective response rate (cORR) of 89.8%, an intracranial cORR of 76.5%, a median duration of response (mDOR) of 49.7 months, and a median progression-free survival (mPFS) of 46.1 months.
- TKI-pretreated: Durable responses were also reported in patients who had previously received a TKI.
- Despite inhibitory effects of taletrectinib on the tropomyosin receptor kinase B (TRKB), neurologic treatment emergent adverse events (TEAEs)* were infrequent (dizziness, 21%; dysgeusia, 15%) and mostly grade 1.
*The integrated safety population includes TKI-naïve and TKI-pretreated patients with ROS1+ NSCLC who received ≥1 dose(s) of taletrectinib 600 mg QD from Phase 2 trials (TRUST-I and TRUST-II) and a Phase 1 trial (J102)
- Several potent TRKB inhibitors, including repotrectinib, entrectinib, and larotrectinib, are associated with significant rates of neurological adverse events (AEs).
- Neurologic AEs related to TRKB inhibitors, such as cognitive impairment, dizziness, ataxia, and peripheral neuropathy, are undesirable; however, a recent publication by Camidge et al hypothesizes that the potential benefit of CNS protection from TRKB inhibition may outweigh these AEs.3
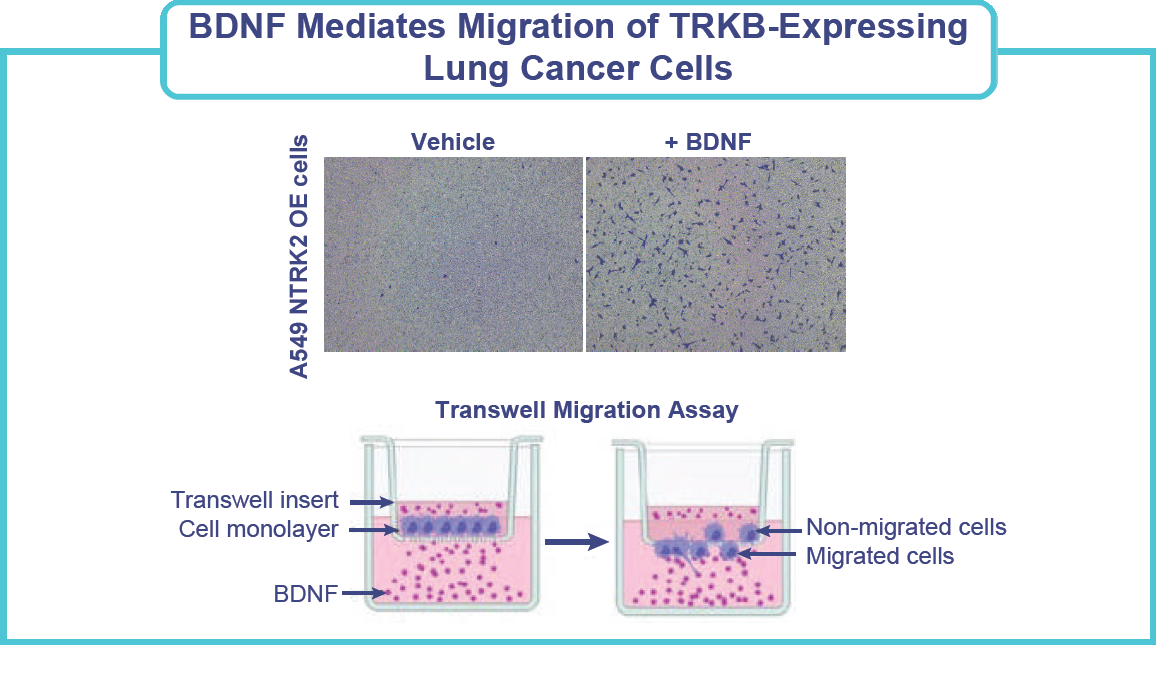
- BDNF-TRKB overexpression and signaling has been associated with increased processes involved in carcinogenesis, such as invasion, migration, epithelial-mesenchymal transition (EMT), metastasis, resistance to cell death due to loss of adhesion (anoikis), and activation of cell proliferation pathways.4
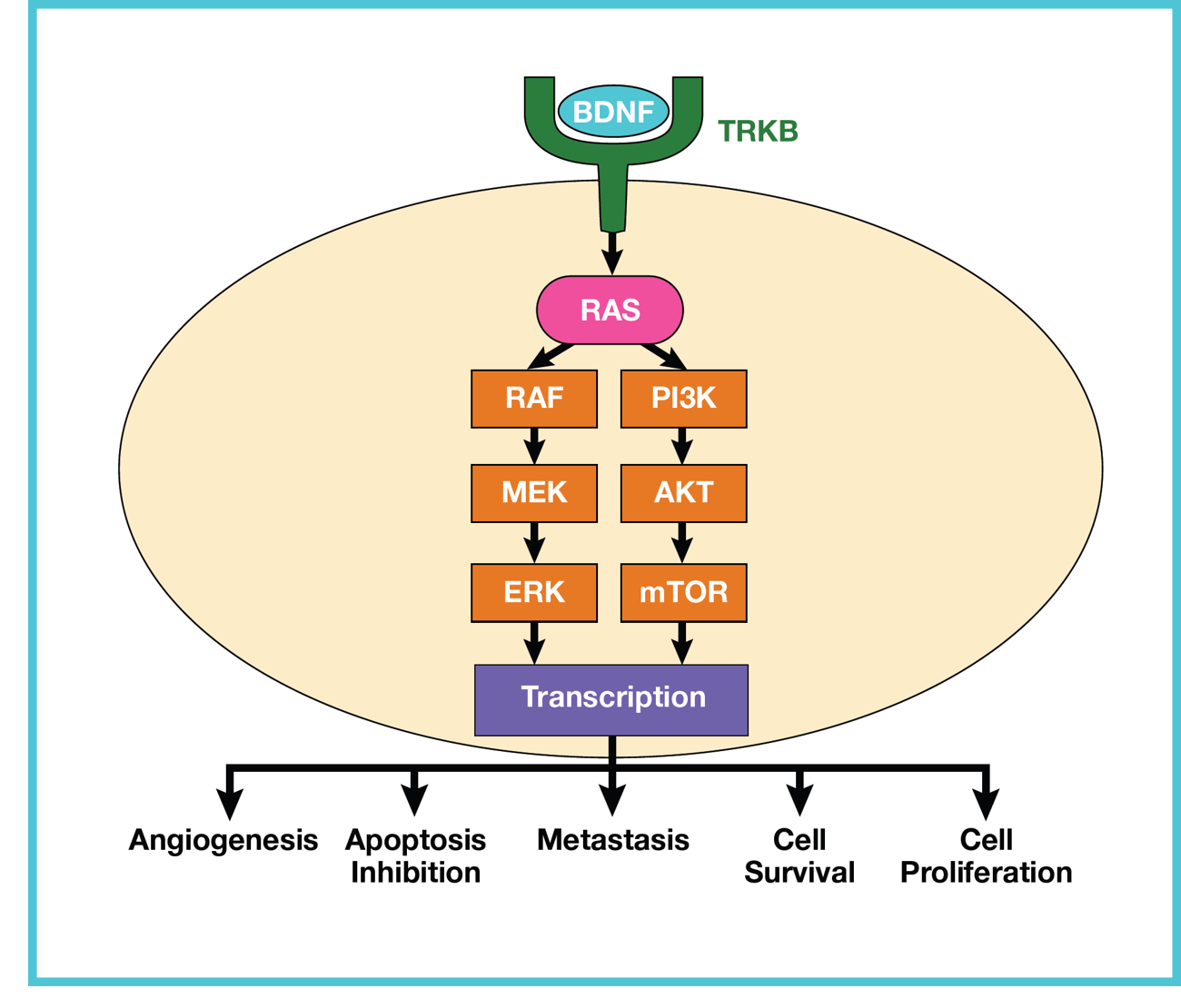
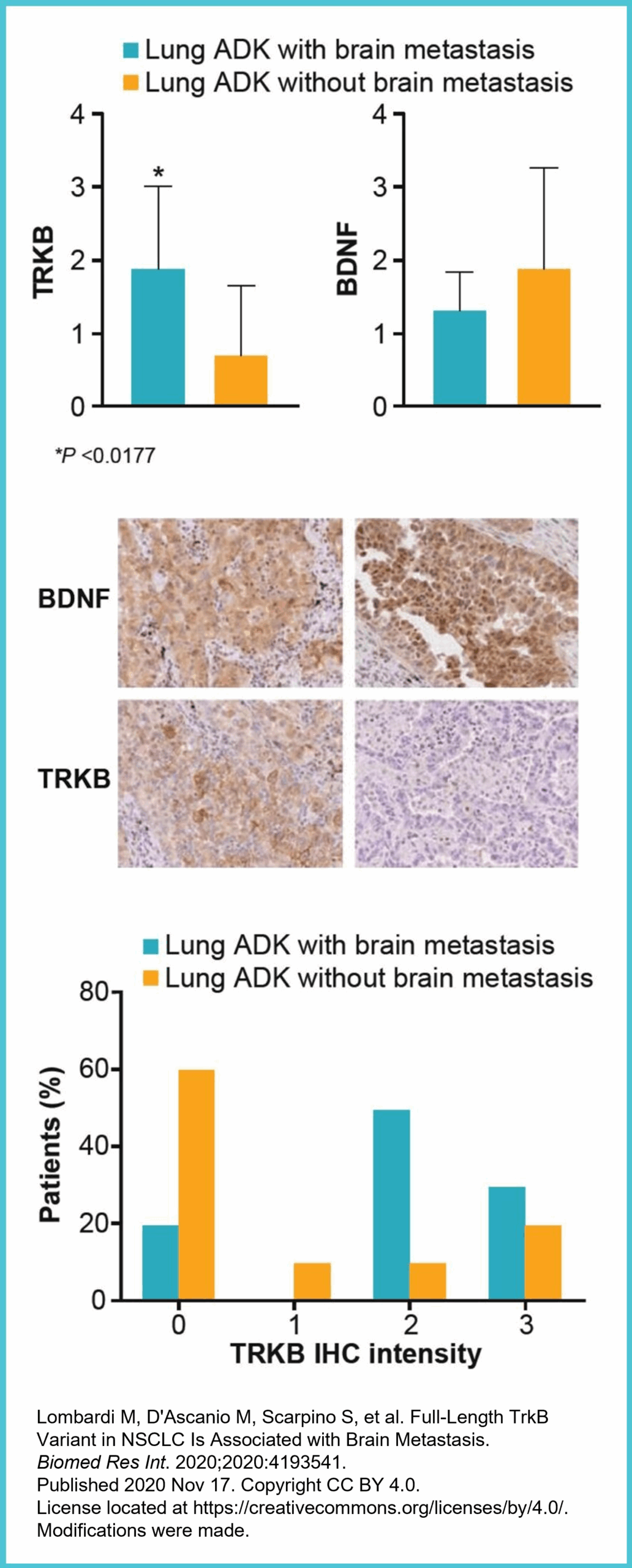
- A retrospective study showed that 80% of lung adenocarcinomas that metastasize to the CNS expressed TRKB receptor, compared with 33% without CNS metastasis.5
- There was a statistically significant increase in the TRKB protein expression in tissue samples obtained from adenocarcinomas (ADKs) of the lung obtained from lung cancer patients with brain metastasis versus patients without brain metastasis.5
- A retrospective study showed that 80% of lung adenocarcinomas that metastasize to the CNS expressed TRKB receptor, compared with 33% without CNS metastasis.5
- Recent research indicates that blocking the BDNF-TRKB signaling pathway, which regulates cancer-neuron synapses, significantly inhibits primary brain tumor progression.3,6
- BDNF-TRKB signaling regulates formation of neuron to NSCLC and breast cancer synapses which are crucial for metastatic CNS progression.3
- Given these findings, the TRKB inhibition profile of ROS1 TKIs such as taletrectinib could be clinically valuable in suppressing CNS progression.3
- ROS1 TKIs lacking TRKB inhibition may not provide the clinical value of CNS protection demonstrated in preclinical breast cancer and clinical NSCLC data.
Objective
Evaluate potential differentiated TRKB inhibition profiles of ROS1 TKIs and their effects on the migratory capacity of lung cancer cells.
Methods
- Cell lines: A549 lung adenocarcinoma epithelial cells were transduced with lentivirus encoding NTRK2 or non-targeting (NT) control. 48 hours after infection, cells were subjected to selection with 0.8 µg/mL puromycin to establish stable pools. Successful overexpression was verified by Western blot analysis.
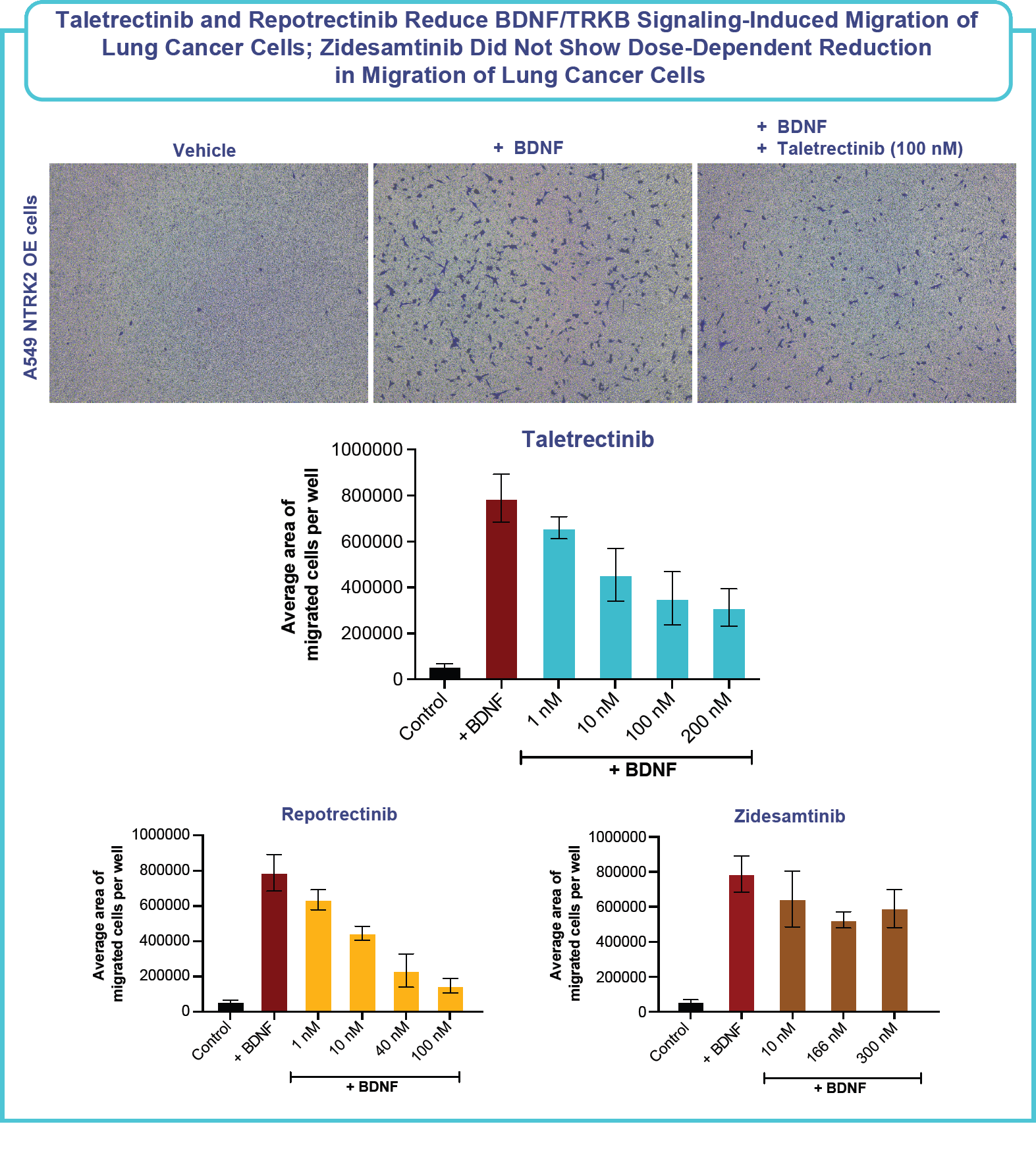
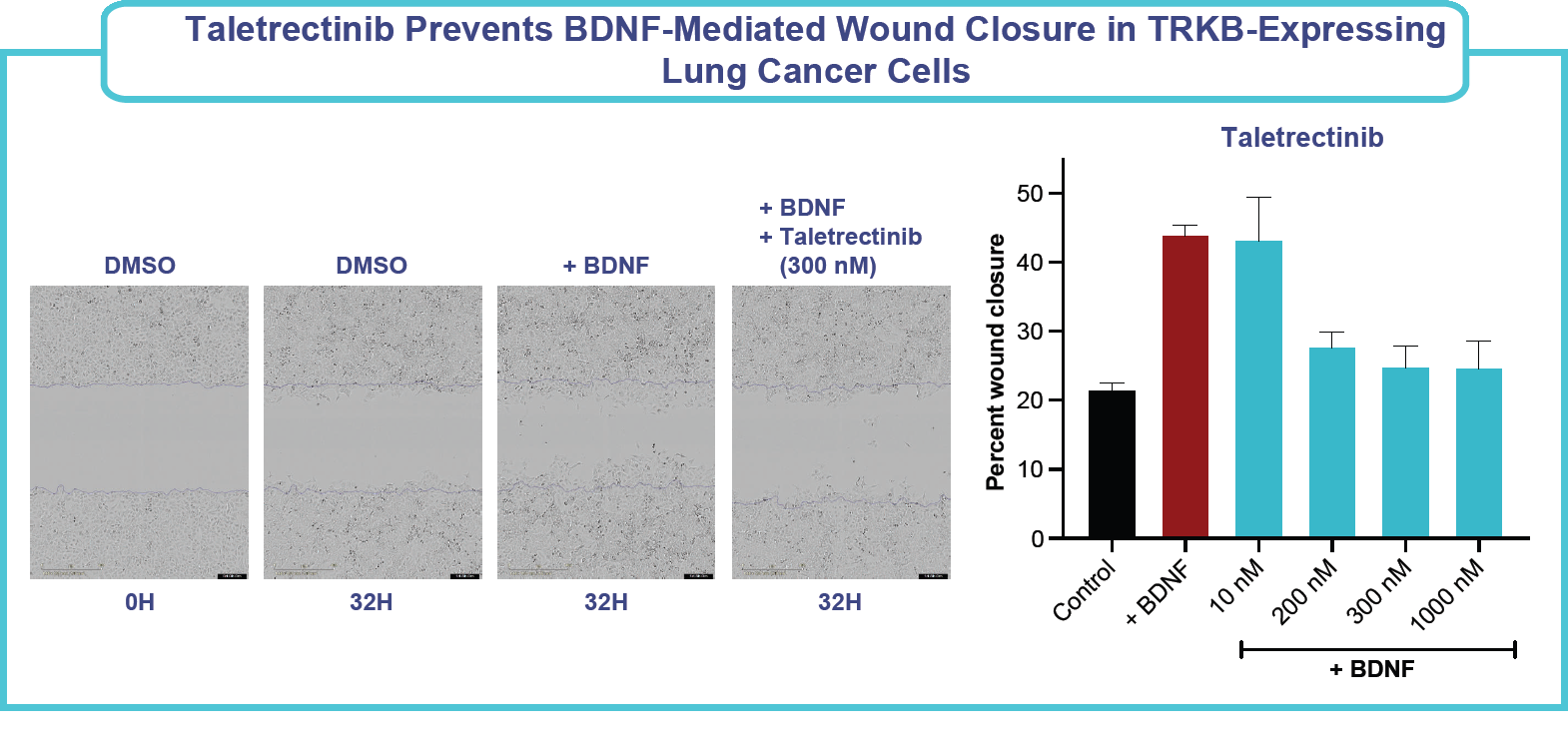
- Migration assay: Migration assays were performed in Transwell plates in accordance with the manufacturer’s instructions (Corning). A549 NTRK2 OE cells were seeded in medium in the top chamber of a Transwell plate along with the test compounds. Serum-free medium supplemented with 50ng/mL BDNF was added to the bottom chamber along with the test compounds. After 24 hours of incubation, nonmigrated cells were removed from the upper insert, and migrated cells on the underside of the insert were fixed using methanol and stained with 0.5% crystal violet solution. Five 10X images of each Transwell insert were taken using an inverted microscope with a camera. ImageJ software was used to count the number of crystal violet-stained cells in each image.
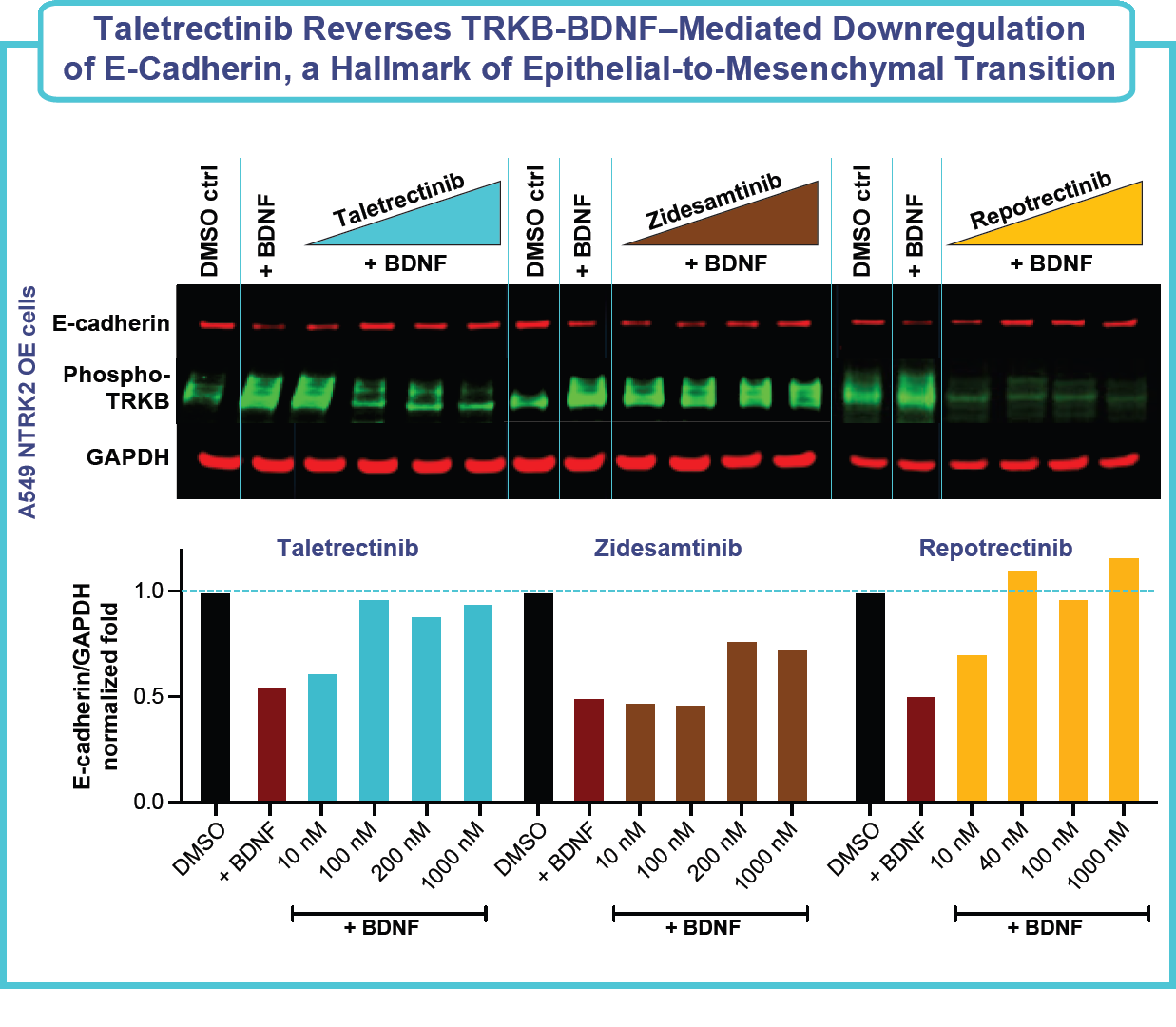
- Western blots: Cells were harvested 48 hours postdosing, and protein expression was analyzed using the antibodies Phospho-TRKA (Tyr674/675)/TRKB (Tyr706/707) (C50F3) (CST-4621), TRKB (CST-4603), E-cadherin (BD610181), and GAPDH (Proteintech_60004); protein expression was normalized to GAPDH expression.
- Intracranial model: CDX studies were run as per standard practice. Briefly, cells were implanted intracranially in mice, and mice were housed in pathogen-free housing with access to sterilized food and water ad libitum. Survival of mice on study was evaluated
Results
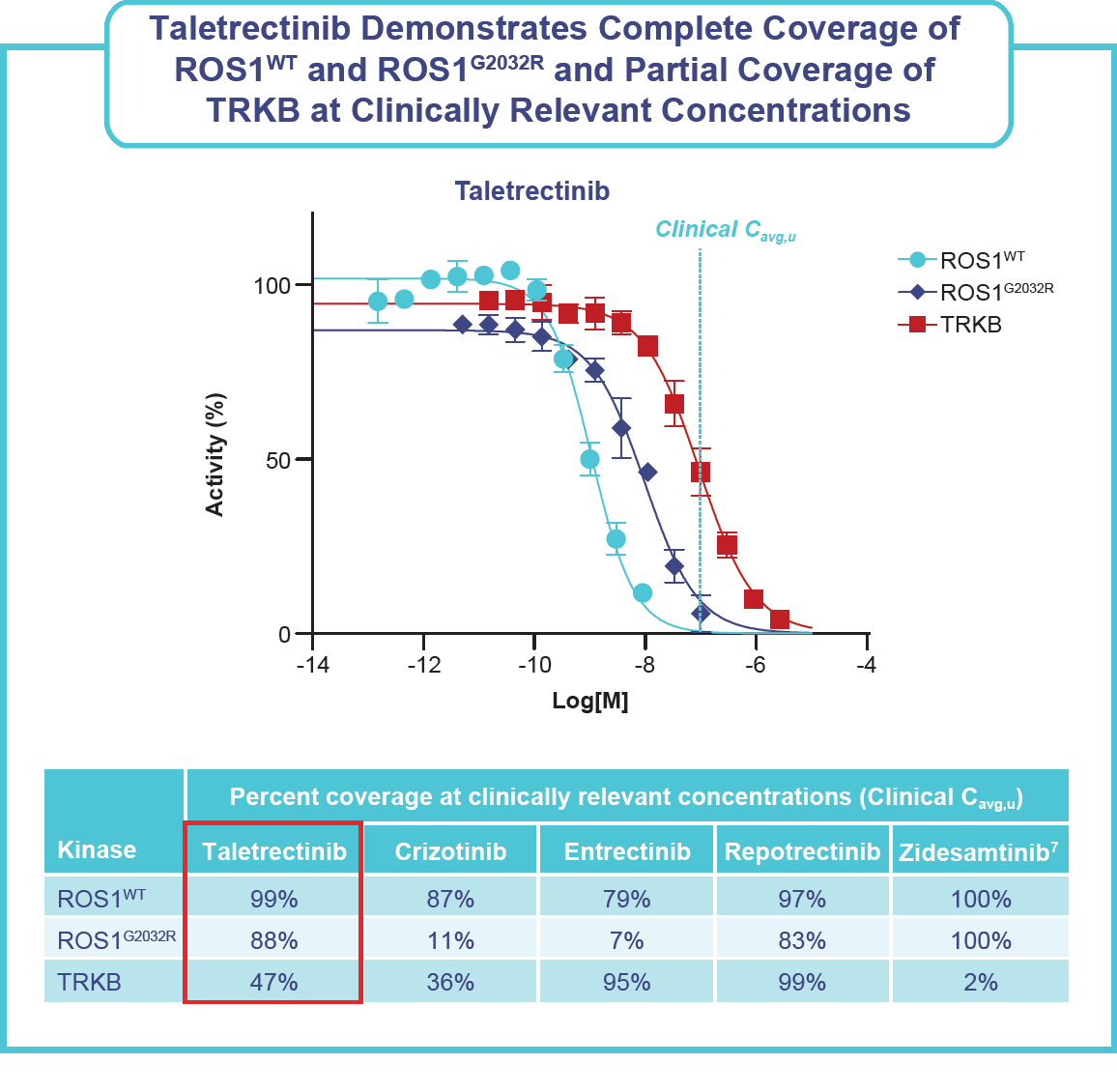
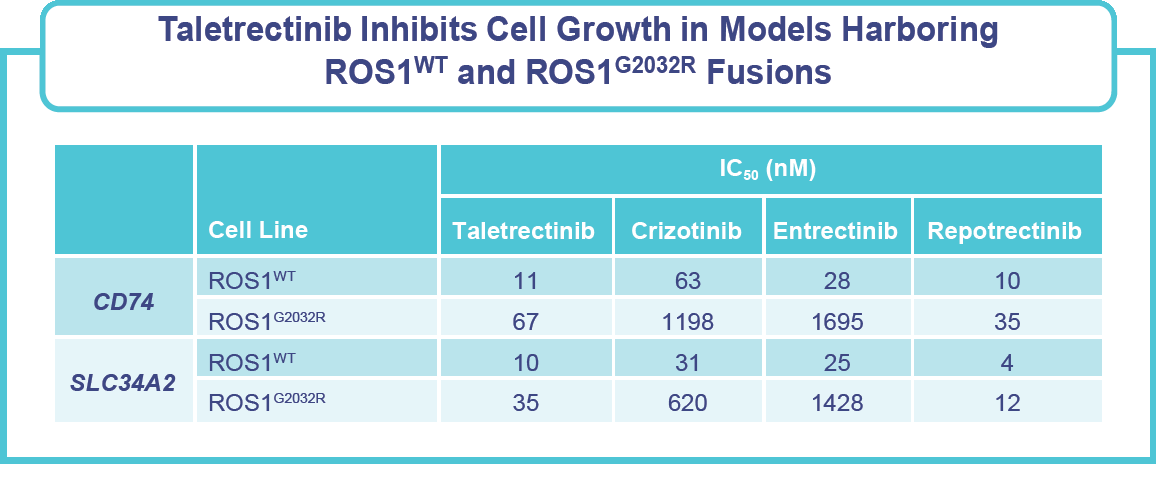
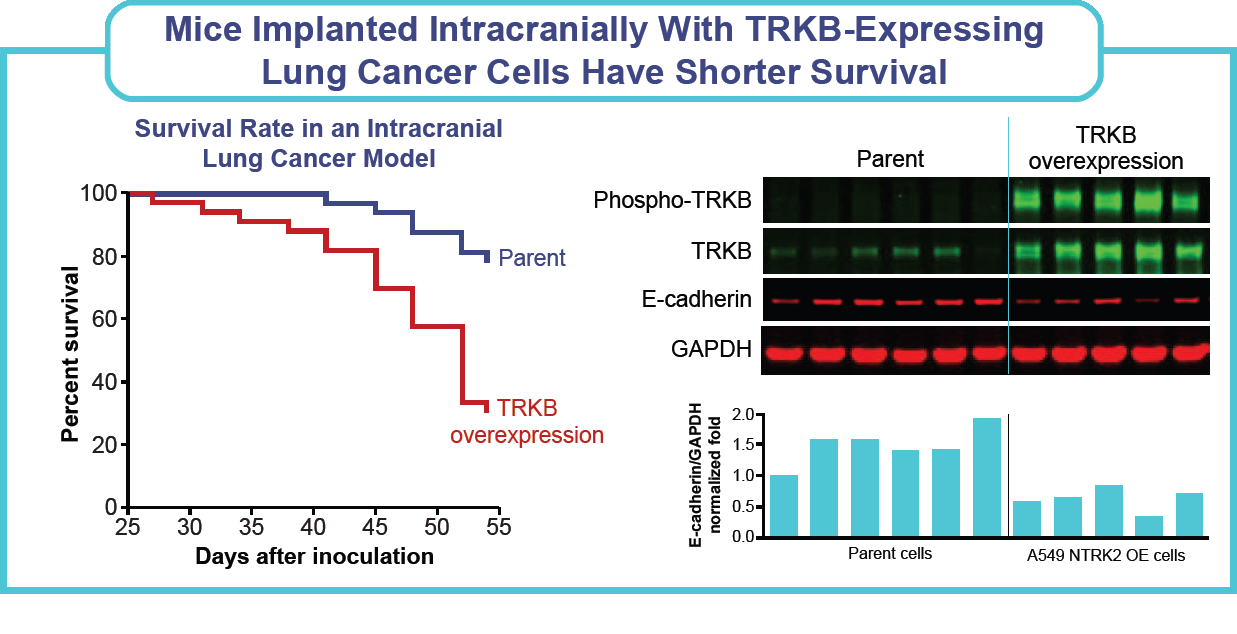
Conclusions
- At clinically relevant concentrations, taletrectinib demonstrates complete coverage of ROS1WT and ROS1G2032R, and partial coverage of TRKB.
- Mechanistic studies revealed that taletrectinib inhibited expression of key markers associated with the EMT pathway and inhibited the migration of lung cancer cells, indicating that taletrectinib’s TRKB activity could potentially reduce the invasive capacity of lung cancer cells.
- Additionally, our nonclinical data showed that zidesamtinib, a TRKB-sparing agent, did not show a dose-dependent reduction in the migration of lung cancer cells and may ultimately lack the potential CNS-protectant effects of TRKB inhibition.
- Taletrectinib’s TRKB inhibition profile diminishes the risk of neurological AEs, while also reducing TRKB-mediated migratory and invasive properties of lung cancer cells, positioning it as a favorable treatment option.3
Abbreviations
ADKs, adenocarcinomas; AE, adverse event; BDNF, Brain-derived neurotrophic factor; Cavg, u, average unbound concentration; CDX, cell line–derived xenograft; CNS, central nervous system; cORR, confirmed objective response rate; ctrl, control; DMSO, dimethyl sulfoxide; DOR, duration of response; m, median; EMA, European Medicines Agency; EMT, epithelial to mesenchymal transition; GAPDH, glyceraldehyde 3-phosphate dehydrogenase; NT, non-targeting; NSCLC, non-small cell lung cancer; OE, overexpression; PFS, progression-free survival; QD, daily; ROS1, proto-oncogene tyrosine-protein kinase 1; TEAE, treatment-emergent adverse event; TKI, tyrosine kinase inhibitor; TRK, tropomyosin receptor kinase; TRKB, tropomyosin receptor kinase B; WT, wild-type.
References
- Boulanger MC, et al. Oncologist. 2024;29:943-956.
- Pérol M, et al. J Clin Oncol. 2025;43(16):1920-1929.
- Camidge DR, et al. J Thorac Oncol. 2026 Jan 20:S1556-0864(25)03067-9.
- Serafim Junior V, et al. Cytokine. 2020;136:155270.
- Lombardi M, et al. Biomed Res Int. 2020;2020:4193541.
- Taylor KR, et al. Nature. 2023;623(7986):366-374.
- Tangpeerachaikul A, et al. Poster presented at: European Society of Medical Oncology Congress 2024; September 13-17, 2024; Barcelona, Spain. Poster #4811
Acknowledgments
- This study was sponsored by Nuvation Bio, Inc.
- Writing and graphical support were provided by Kelsi Bates, PharmD, from Phillips Group Oncology Communications, Inc. (Albany, NY, USA), and funded by Nuvation Bio

